Complex Scenarios in Advanced Melanoma: Examining Three Patient Cases
Despite significant developments on the advanced melanoma treatment front, “many clinical scenarios remain where there is no clear-cut course of action," said Robert H.I. Andtbacka, MD, CM, during a recent OncLive Peer Exchange®.
Robert H.I. Andtbacka, MD, CM

Robert H.I. Andtbacka, MD, CM
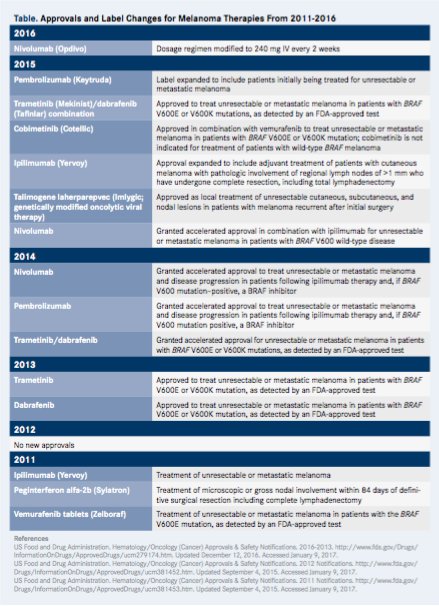
The number of treatment options for advanced melanoma continues to increase steadily. Since March 2011, when ipilimumab (Yervoy) was the first new drug approved by the FDA for melanoma in 13 years, many agents and combination therapies have come to market or received expanded indications or other label changes (Table).1-3 Many other treatments continue to show promise in clinical trials. Yet despite significant developments on the advanced melanoma treatment front, “many clinical scenarios remain where there is no clear-cut course of action,” said Robert H.I. Andtbacka, MD, CM, during a recent OncLive Peer Exchange®.
During the Peer Exchange, Andtbacka used 3 case scenarios to lead a panel of melanoma experts in a discussion of some of the key challenges impacting clinical practice today. The cases consisted of an elderly adult with newly diagnosed BRAF wild-type stage IV melanoma, a patient with BRAF-mutant stage IV melanoma, and a patient with recurrent disease after resection.
A key area of discussion for this case scenario focused on molecular testing. In addition to testing for BRAF, the mutational status of 2 other genes were noted to be of potential interest: NRAS and the c-Kit oncogene. “NRAS is the second most common oncogenic mutation we see in cutaneous melanoma patients, and, in part, it’s because the NRAS and BRAF V600 mutations are mutually exclusive. Seeing an NRAS mutation increases our confidence in the specificity and accuracy of a wild-type BRAF test,” said Michael A. Davies, MD, PhD. Although these mutations are “quite rare,” he explained that his interest in testing for these mutations stems from an availability of clinical trials for patients with NRAS mutations and clinical evidence that patients with KIT mutations, which occur more commonly with mucosal melanomas, can benefit from treatment with KIT inhibitors.
More evidence and standardization are needed, however, before NRAS and KIT mutations become routine testing targets in clinical practice. The panelists noted the same to be true for PD-L1 expression testing. “In melanoma, nivolumab and pembrolizumab do not require testing for PD-L1 expression,” said Antoni Ribas, MD, PhD, whereas initiation of BRAF/MEK inhibitor therapy requires BRAF V600E or V600K testing, he explained. Furthermore, although PD-L1 expression has been associated with treatment response, it is not a definitive test.
Case 1 - BRAF Wild-type Stage IV Melanoma

“When you look at the receiver operating characteristic curve, there is no optimal cutoff; the test is not sensitive and it is not specific to predict response and progression-free survival [PFS]... so we [only] use it in research to look at the full tumor in the microenvironment,” said Georgina Long, MD, BSc, PhD, MBBS. Nevertheless, as testing evolves and more data become available, mutation testing is expected to become more comprehensive. “In the future, we’re going to be testing for as many mutations as possible. The techniques are already there, but their standardization and who pays for them is limiting our routine use of them,” said Ribas.
In terms of treatment options, the panelists agreed that PD-1 inhibition would be an optimal approach for this patient, whether as single-agent therapy or as a combination therapy with ipilimumab. “For this patient with very low-volume metastatic disease involving the subcutaneous tissues and lung, I’d feel very comfortable starting her on a single-agent PD-1 inhibitor, such as nivolumab or pembrolizumab, as a first-line treatment approach,” said Michael A. Postow, MD. “We’ve known that patients with small lung metastases can do very well with single-agent PD-1, and we don’t yet have a perfect predictive set of clinical variables or any kind of correlative variables that tell us who exactly can benefit from PD-1 monotherapy versus who may need the combination,” he said.
Although nivolumab/ipilimumab combination therapy has shown a higher response rate than nivolumab monotherapy, it has a much higher toxicity rate, which can be a limiting factor, particularly in more vulnerable or frail patients. Thus, the panelists largely agreed that more aggressive combination immunotherapy might be better reserved for patients with signs of more aggressive disease, such as a rapid increase in tumor size or elevated serum lactate dehydrogenase (LDH) levels.
Regardless of treatment approach, “one of the keys in a 75-year-old patient that you would want to know is how hard would it be for this patient to be brought in for medical care if the patient were to get ill,” said Davies. The panelists concurred that age alone should not be considered a contraindication to using a potentially more toxic regimen when it is needed, but that more careful planning is warranted in such cases to improve outcomes in the event adverse effects (AEs) occur.
Case 2 - BRAF Wild-type Stage IV Melanoma
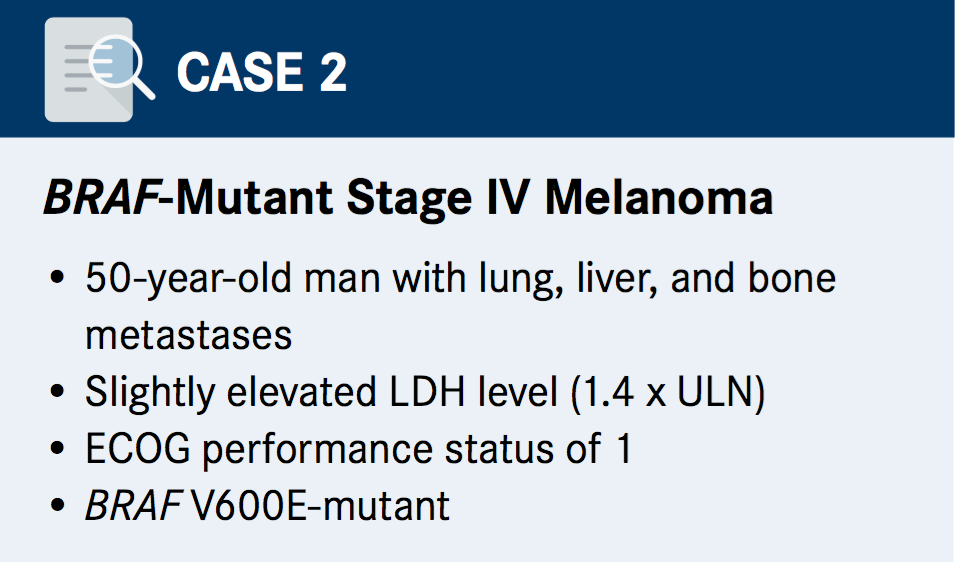
A key area of focus this second case was on treatment and whether BRAF/MEK inhibitors are preferable to combination immunotherapy. “In Australia, we have funding rules, which means we must give a BRAF and MEK inhibitor for BRAF-mutant patients in the frontline setting no matter what their disease state is,” said Long. “We actually don’t agree with that, and things may be changing in some respects in the next few months, but I’ve tended to try and find immunotherapy clinical trials for my patients.”
She said if she could choose a frontline immunotherapy in this setting, she’d select combination immunotherapy with nivolumab and ipilimumab, which has shown more robust response rates than other currently available treatments. However, this remains a data-free zone.
“At this point, we really don’t have any head-to-head trials for patients with metastatic BRAF-mutant melanoma showing what is the right initial therapy,” said Davies, noting that this is currently being evaluated in clinical trials. “Patients like [the case patient] are being randomized to start with a BRAF and MEK inhibitor combination therapy versus combination immunotherapy with ipilimumab and nivolumab, and if they progress, are crossed over to the other therapy,” he explained. “This is really going to provide us with the definitive information about which of those therapeutic approaches is the right one to start with,” he said, noting that trials are also currently underway assessing combining targeted therapy with immunotherapy.
Table - Approvals and Label Changes for Melanoma Therapies From 2011-2016
Based on currently available evidence, patients who progress on immunotherapy can respond to subsequent targeted therapy and vice versa, said Ribas, although the response appears to be lower when immunotherapy is initiated after progression on targeted therapy. Nevertheless, it remains a viable option and he suggested that treatment decisions should continue to be personalized to patients’ circumstances and preferences. “Even when we have randomized data, we can only tell what would happen to most patients, but it will never tell us what will happen to an individual patient, so we’ll all continue to discuss and personalize therapy,” he said.
When personalizing treatment, many key factors can impact decision making. One critical factor in this case patient’s scenario is his elevated LDH, which is a factor that has been associated with worse outcomes and a lower response to immunotherapy, though responses can be durable when they occur.
“For patients with a high LDH, we know the targeted therapies have very high response rates, probably higher than what we can achieve with immunotherapy, but the overwhelming majority of those responses have short duration,” said Davies. However, because the case patient’s LDH is only slightly elevated and he is not highly symptomatic, there is more flexibility in time to achieve response than in a highly symptomatic patient with markedly elevated LDH and high disease burden. This enables other key decision-making factors to bear more weight, such as the patient’s ability and willingness to receive immunotherapy infusions every 2 to 3 weeks in the clinic versus orally taking a targeted agent at home.
Despite infusions being more burdensome upfront, Postow noted that they are not always a long-term burden. “At least with combination ipilimumab and nivolumab, a lot of patients discontinue treatment after just a few infusions upon achieving response, particularly in the setting of toxicity, and many of those patients are not on ongoing treatment longer term...so there is something to be said for the long-term PFS you can get from these short durations of treatment in some patients,” he said.
At the same time, the panelists made clear that frontline BRAF inhibitor-based therapy does not preclude long-term survival, particularly in patients with normal LDH and 3 or fewer sites of metastasis. “The 3-year overall survival is well above 50% to 60% for these patients,” said Long, with data also showing favorable PFS. More data will become available as clinical trials mature. “Once we start getting out to 3, 4, 5 years and see the pure activity of the drugs, we’ll be able to have better conversations about upfront treatments,” she said.
Case 1 - Recurrent Disease After Resection

This case scenario was the most controversial of the 3 because it covers an area where there are more limited data regarding the role of systemic treatments. The panelists agreed on resection being appropriate for the patient, but data on whether to use neoadjuvant or adjuvant therapy in such cases remains a murky area that still requires 2 key questions to be more definitively answered: (1) will systemic therapy reduce the risk of such high-risk patients developing distant metastatic disease; and (2) will using systemic therapy upfront in patients with large or impinging tumors sufficiently reduce tumor burden to improve post-operative morbidity.
When neoadjuvant therapy is being pursued, it must be done in a clinical trial setting, the panelists noted. “Neoadjuvant would not be done outside of a clinical trial and surgery would be done upfront if we did not have a trial for this patient...and then discuss an adjuvant trial,” said Long. In contrast, adjuvant systemic therapy could be done outside of a clinical trial. “[In the clinical setting], we would be discussing high-dose ipilimumab or high-dose interferon,” said Ribas, who later said pegylated interferon could be another option for US patients, although it does not appear to offer an advantage over high-dose interferon in terms of toxicity or survival.
Compared with high-dose ipilimumab, high-dose interferon has significantly more toxicity, with most patients experiencing some AEs. “It’s given intravenously for 5 days on, 2 days off for 1 month, and during that time, most patients have fevers, chills, and malaise, with treatment often needing to be stopped because of pancytopenia or elevated liver function tests,” said Ribas. “Then patients continue with subcutaneous dosing 3 times a week for 1 year, where they would continue feeling tired most of the time, and they can also develop depression because of the drug,” he said. Although most patients receiving high-dose ipilimumab will not develop AEs, when they occur, they can be severe and include complications like colitis, hypophysitis, and thyroiditis, he said.
In patients with metastatic disease, ipilimumab is dosed at 3 mg/kg, but in the adjuvant setting, a dose of 10 mg/kg has been approved. “In the community, we are still seeing patients being treated with 3 mg/kg with really no data to support that,” said Andtbacka.
Current evidence suggests a dose-response effect with ipilimumab, with improved outcomes in patients receiving the 10 mg/kg dose versus the 3 mg/kg dose, which has led to a dosing conundrum because patients with more serious metastatic disease are being treated with lower doses of ipilimumab than patients in the adjuvant setting who may already be cured. The challenge with the higher dose, however, is significantly more toxicity, the extent of which still needs to be teased out in clinical trials.
“My standard is giving the 10 mg/kg dose and hoping that the patient will tolerate it,” said Ribas. In the meantime, he said the 3 mg/kg is being tested prospectively in the ECOG1609 trial (NCT01274338), which is comparing high-dose interferon, ipilimumab 10 mg/kg, and ipilimumab 3 mg/kg in patients with high-risk stage III to IV melanoma that has been excised. Study results are anticipated this year.
The panelists agreed that biochemotherapy and radiation therapy would not be appropriate treatment options for the case patient. “At this point, the data have shown no impact on overall survival [in the adjuvant setting], so I would not recommend biochemotherapy,” said Davies. He encouraged enrolling patients into clinical trials to improve the adjuvant therapy landscape for earlier stages of disease.
When considering radiation, the panelists agreed that large groin tumors are not ideally suited to this approach either. “Our experience with the groin is that the risk of having infections, lymphedema, and AEs most of the time outweighs the potential benefit, so we have really decreased the amount of radiation that we do to the groin,” said Andtbacka. He said that vast improvements in systemic treatments have also helped move treatment away from radiating groin lesions.
In their concluding statements, the panelists concurred that treatment decisions always need to be personalized based on the best and most current data available and on the patient’s preferences and tumor characteristics. “How we use our existing therapies most appropriately remains a challenge, but it’s a challenge that’s much more pleasant now that we have better therapies,” said Davies. The panelists all agreed that the melanoma armamentarium is only going to continue to expand in the coming years. The hope is that this will help resolve some challenges and lead to a more acceptable melanoma landscape.




