
Neoadjuvant pembrolizumab plus chemotherapy led to an improvement in pathological complete response rate compared with chemotherapy plus placebo in patients with high-risk, early-stage, estrogen receptor–positive, HER2-negative breast cancer.

Your AI-Trained Oncology Knowledge Connection!


Neoadjuvant pembrolizumab plus chemotherapy led to an improvement in pathological complete response rate compared with chemotherapy plus placebo in patients with high-risk, early-stage, estrogen receptor–positive, HER2-negative breast cancer.

The European Commission has approved sacituzumab govitecan-hziy for the treatment of adult patients with unresectable or metastatic hormone receptor–positive, HER2-negative breast cancer who have received endocrine-based therapy and at least 2 additional systemic therapies in the advanced setting

New agents in development including ABBV-011 and BL-B01D1 seek to turn the tides of relapsed extensive-stage small cell lung cancer; although chemoimmunotherapy has earned its right as the frontline standard of care, both classes of agents have yet to demonstrate benefit in second or later lines.

Bhavana “Tina” Bhatnagar, DO, discusses the mechanism of action of uproleselan, the potential efficacy and safety benefits of adding this agent to standard therapeutic regimens in patients with AML, and the importance of considering MRD negativity as an end point in AML clinical trials.

The University of Alabama at Birmingham, in partnership with the Alabama Department of Public Health and the American Cancer Society, recently launched Wipe Out Cervical Cancer — a statewide action plan to eliminate cervical cancer as a public health problem in Alabama.

Long-term data demonstrated that treatment with cosibelimab improved complete response rates over time in patients with locally advanced or metastatic cutaneous squamous cell carcinoma in a phase 1 trial.

The European Commission has approved piflufolastat (18F) for the detection of prostate-specific membrane antigen–positive lesions with positron emission tomography in adults with prostate cancer.

Addressing socioeconomic health disparities has been a crucial objective of health policy in the United States. The relationship between cancer incidence and mortality and socioeconomic status varies across malignancies.
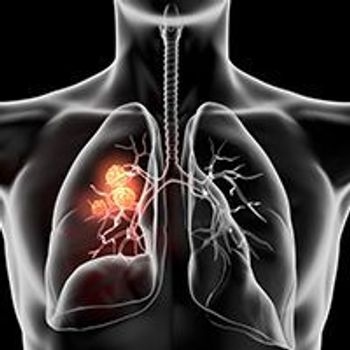
Neoadjuvant treatment with nivolumab plus platinum-based chemotherapy followed by adjuvant nivolumab increased pathological complete response rates and prolonged survival compared with neoadjuvant chemotherapy alone in patients with resectable stage IIIA/B non–small cell lung cancer.

Positive event-free survival data from the phase 3 AEGEAN, NEOTORCH, and KEYNOTE-671 trials add further evidence to the benefit of perioperative immunotherapy in early-stage non–small cell lung cancer but have yet to show clear biomarkers of response beyond PD-L1.

Overall survival analysis from ADAURA bolsters the already impressive disease-free survival data for patients with EGFR-mutant non–small cell lung cancer.

Sarah E. S. Leary, MD, MS, spotlights the importance of developing effective treatments for pediatric patients with low-grade glioma, key findings with tovorafenib in patients with RAF mutations, and how this agent may influence the low-grade glioma therapeutic landscape going forward.

Pasi A. Jänne, MD, PhD, details common EGFR mutations in NSCLC, the importance of testing for EGFR mutations in all patients, the clinical implications of overall survival data for adjuvant osimertinib from the ADAURA trial, and ongoing trials examining combination therapies to potentially improve upon the use of single-agent osimertinib.

Press Release
The platinum drugs, carboplatin and cisplatin, known for effectively treating lung, head and neck, breast, ovarian, gastric, bladder and testicular cancers, have been in short order for the past few months.

Treatment with UGN-102 with or without transurethral resection of bladder tumors led to an improvement in disease-free survival compared with TURBT alone in patients with low-grade, intermediate-risk non–muscle-invasive bladder cancer.

The phase 1/2 UPLIFT trial evaluating the antibody-drug conjugate upifitamab rilsodotin in patients with platinum-resistant ovarian cancer failed to meet its primary end point of investigator-assessed overall response rate in the NaPi2b-positive population.

The implementation of a pharmacist-driven biosimilar substitution program across American Oncology Network institutions resulted in increased uptake of biosimilar agents as well as financial savings for payers, patients, and providers.

The allogeneic natural killer cell therapy NKX019 was found to have an acceptable toxicity profile and to induce deep and durable responses in patients with relapsed or refractory B-cell non-Hodgkin lymphomas.

Isabelle Franklin discusses the effects of social determinants of health on rates of colorectal cancer screening, expands on how these social determinants of health could affect a patient’s likelihood of adhering to screening guidelines, and highlights ways to potentially address these barriers to screening.

Erika P. Hamilton, MD, discusses the role of trastuzumab deruxtecan in HER2-positive and HER2-low breast cancer; the evolution of CDK4/6 inhibitors in hormone receptor–positive disease; and how genetic testing can match patients with personalized therapies and support further precision medicine research.
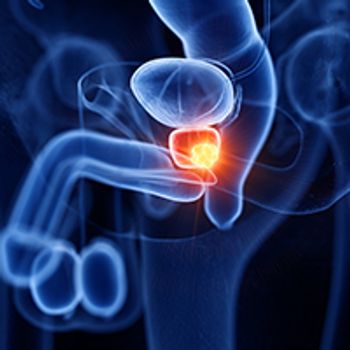
Varied levels of compliance were observed in terms of using validated quality indicators to determine the quality of active surveillance for patients with low-risk prostate cancer, according to findings from a population-based study published in the Journal of the Comprehensive Cancer Network.

The combination of mRNA-4157 and pembrolizumab will be further evaluated as an adjuvant treatment option for patients with resected, high-risk, stage IIB to IV melanoma in the phase 3 V940-001 trial.

Press Release
Rutgers Cancer Institute of New Jersey, the state’s only National Cancer Institute-designated Comprehensive Cancer Center together with RWJBarnabas Health, has treated its first patient using genetically modified T-cells that were manufactured in its own state-of-the-art Good Manufacturing Practices facility, a fully commissioned clean space for manufacturing of viral vectors and cell products for human administration.

The National Comprehensive Cancer Network has added flotufolastat F 18 injection to its Clinical Practice Guidelines in Oncology for positron emission tomography of prostate-specific membrane antigen–positive lesions in men with prostate cancer with suspected metastasis who are candidates for initial definitive therapy.

The FDA has issued a ‘safe to proceed’ for the investigational new drug application enabling a phase 1 study evaluating NY-ESO-1 TCR/IL-15 NK, a first-in-class engineered T-cell receptor natural killer cell therapy for patients with relapsed/refractory multiple myeloma.

Findings from an analysis of treatment patterns for patients with systemic amyloid light chain amyloidosis have demonstrated that several unmet needs exist, including timely diagnosis and limited survival outcomes for patients with advanced disease.
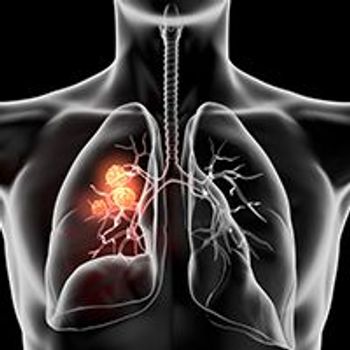
Sugemalimab plus chemotherapy demonstrated statistically significant and clinically meaningful improvements in progression-free survival and overall survival vs placebo plus chemotherapy for the first-line treatment of patients with stage IV non–small cell lung cancer irrespective of PD-L1 expression level and tumor pathological type.

Selinexor maintenance generated an improvement in progression-free survival compared with placebo in patients with TP53 wild-type advanced or recurrent endometrial cancer who responded to first-line chemotherapy.

ASCO has released an update to the guidelines for the practical assessment and management of vulnerabilities in older patients receiving systemic cancer therapy.

Angel Qin, MD, contextualizes the non–small cell lung cancer ADAURA trial, in which patients with EGFR-mutated non–small cell lung cancer who received adjuvant therapy with osimertinib achieved a 5-year overall survival rate of 85% vs 73% in those who received placebo.