
The technological advances in analyzing the human genome have spawned a new era in breast cancer as well as other types of malignancies that will affect oncology practice and will necessitate dramatic changes in the clinical trials system.

Your AI-Trained Oncology Knowledge Connection!


The technological advances in analyzing the human genome have spawned a new era in breast cancer as well as other types of malignancies that will affect oncology practice and will necessitate dramatic changes in the clinical trials system.

The strategy of targeting aberrant signaling in the JAK pathway appears promising thus far for SAR302503, an oral agent in development for the treatment of myelofibrosis.

An interview with Alice Tsang Shaw, MD, PhD, who has won broad recognition for her contributions to the treatment of NSCLC, including the use of crizotinib to target the ROS1 gene rearrangement and research into other emerging agents.

A study of patients newly initiated to erlotinib therapy for NSCLC found that there is no uniformity in the billing codes used for pharmacogenomic testing prior to starting the drug, making it difficult to analyze treatment patterns and cost impact.

Jeanette H.W. Leusen, PhD, focuses on studying the working mechanisms of therapeutic antibodies and the biology of fragment crystallizable receptors, including the anti-CD20 monoclonal antibody rituximab in patients with non-Hodgkin lymphoma.
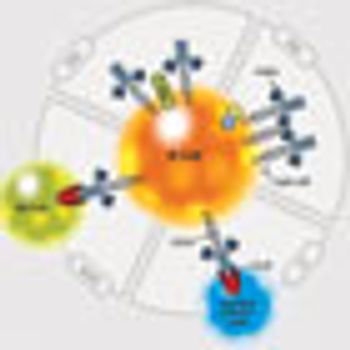
A large proportion of patients become rituximab-refractory, which has prompted the development of newer CD20 agents with altered structures that are designed to improve upon rituximab's performance.

Novel therapies have heightened the need for clinicians to be well informed on potential skin-related adverse events to help ensure that patients adhere to their regimens and complete treatment.
Two recent reports have challenged certain assumptions in the drug development process that raise ethical concerns, individually and collectively, which the clinical cancer research community must address.

Researchers have identified a mechanism that explains why patients with glioblastoma have not had successful outcomes when treated with inhibitors of mTOR despite the fact that it is overexpressed in approximately 90% of cases.