In many circumstances, the absence of so-called definitive evidence does not equate with the absence of evidence, just the level required by some to declare an approach to be an acceptable standard-of-care option.

In many circumstances, the absence of so-called definitive evidence does not equate with the absence of evidence, just the level required by some to declare an approach to be an acceptable standard-of-care option.

Age is no longer a consideration when determining whether an older patient with blood cancer is a candidate for stem cell transplantation.

Amid rapid progress in the treatment of advanced lung cancer, several leading organizations have joined in developing new guidelines for the optimal use of molecular testing to help select patients for therapy with tyrosine kinase inhibitors

It is well recognized that advances in cancer management have most often resulted from a close collaboration between clinical and laboratory investigative efforts.
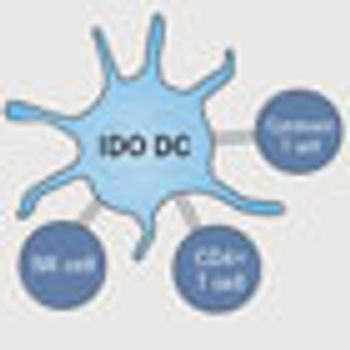
IDO is a key enzyme in the normal regulation of the host's adaptive immune response. Its role in regulating the immune response was initially demonstrated when pregnant mice were given IDO inhibitors, resulting in the rejection of the unborn fetus by the maternal immune system.
Afatinib (Gilotrif) tablets were approved by the FDA on July 15, 2013, as a first-line oral treatment for non– small cell lung cancer with EGFR mutations. The oral tablets became commercially available in the United States September 2.

The list of agents currently available to treat soft-tissue sarcoma is rather short, with no drugs being approved on the basis of an improvement in overall survival in approximately 20 years.

Douglas Hanahan and Robert Weinberg acknowledged the importance of the immune system in cancer development in 2011, when they added immune evasion to their list of "hallmark" abilities that are essential for the transformation of normal cells into cancerous ones.

The optimal use of adjuvant chemotherapy for patients with early-stage non-small cell lung cancer remains unresolved, with several clinical trials under way in Europe aimed at identifying ways to better select patients for treatment

Whether it involves the minutiae of preclinical studies or the broad perspective of group leadership, if the work concerns gynecologic oncology, Ronald D. Alvarez, MD, has likely tried it.

The PAM50 test, designed to determine a risk of recurrence score for patients with breast cancer, adds significant prognostic information to clinical decision making and might be superior to other risk assays.