
Metronomic treatment approaches could represent therapeutic avenues for patients with TP53-mutated myeloid malignancies.

Metronomic treatment approaches could represent therapeutic avenues for patients with TP53-mutated myeloid malignancies.

The CSF-1R–targeting monoclonal antibody emactuzumab met key end points in the phase 3 TANGENT study, setting the stage for a BLA submission.

The allogeneic CAR T-cell therapy cema-cel improved MRD negativity rates vs observation as first-line consolidation in LBCL.
Frontline darovasertib plus crizotinib generated a PFS improvement vs treatment of investigator’s choice in HLA-A*02:01–negative metastatic uveal melanoma.
Sac-TMT plus pembrolizumab was safe and effective in recurrent/metastatic cervical cancer.
Tisotumab vedotin plus carboplatin & pembrolizumab ± bevacizumab achieved 65.8% ORR and 28-month median OS in first-line recurrent/metastatic cervical cancer.

Mirvetuximab soravtansine plus carboplatin showed antitumor activity and a manageable safety profile in patients with recurrent platinum-sensitive ovarian cancer.
Significantly improved response rates were shown for liso-cel vs real-world data for SOC in later-line relapsed or refractory mantle cell lymphoma.
ECOG PS should be evaluated before initiating this agent, as patients with poorer PS may experience shorter life expectancy than reported in trials.

Treatment with daraxonrasib led to PFS and OS benefits vs SOC cytotoxic chemotherapy in patients with metastatic pancreatic ductal adenocarcinoma.

The FDA has accepted and granted priority review to the BLA seeking the approval of I-DXd in ES-SCLC.

Roswell Park Comprehensive Cancer Center researchers are investigating whether CAR T-cell therapy could target some of the most common autoimmune diseases.

Pirtobrutinib plus venetoclax and rituximab improved PFS in relapsed/refractory CLL/SLL, meeting the primary end point of BRUIN CLL-322.
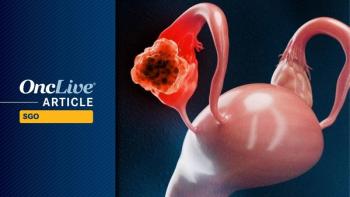
Sofetabart mipitecan elicited strong antitumor activity in recurrent platinum-resistant high-grade serous ovarian cancer.
Ibrutinib given prior to apheresis was associated with improved outcomes with tisagenlecleucel vs postapheresis administration in relapsed/refractory LBCL.

R. Lor Randall, MD, FACS, highlights a recent meta-analysis comparing 2 approaches to surgical management for patients with femoral metastases.
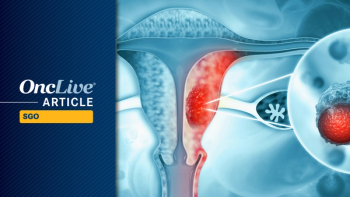
The novel ADC DB-1311/BNT324 was effective in cervical cancer and PROC, regardless of prior treatment or histology

The B7-H3–targeting ADC demonstrated encouraging responses across multiple gynecologic cancers in a phase 1/2 trial.

Single-agent rezatapopt showed efficacy and safety irrespective of patients' platinum status, prior bevacizumab or PARP inhibitor exposure, or FRα expression.
The B7-H4–targeted ADC produced ORRs of 62% and 67% in patients with PROC and endometrial cancer, respectively, in the phase 1 BEHOLD-1 study.

The top 5 OncLive TV videos of the week cover insights in mantle cell lymphoma, chronic lymphocytic leukemia, salivary gland cancer, cholangiocarcinoma, and prostate cancer.

Cadonilimab combined with chemotherapy demonstrated robust efficacy and a favorable safety profile in advanced or recurrent endometrial cancer.

Patients who received first-line niraparib as maintenance therapy had high resolution rates and low number of discontinuations due to hematologic PSEOIs.

A significant increase in treatment completion was observed among Black women with high-risk endometrial cancer who received 1:1 peer support.

Trastuzumab pamirtecan achieved ~50% ORR in pretreated HER2-expressing endometrial cancer, earning FDA breakthrough designation per phase 1/2 trial data.

With 1 year of additional follow-up, avutometinib plus defactinib showed durable efficacy with no new safety concerns in LGSOC.

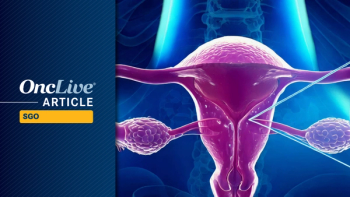
Results from NRG-GY019 establish chemotherapy followed by letrozole as the definitive SOC for newly diagnosed stage II-IV low-grade serous ovarian carcinoma

Abemaciclib plus letrozole was safe and active in advanced/recurrent endometrioid endometrial carcinoma.

Relacorilant plus nab-paclitaxel cuts death risk by 35% and extends survival to 16 months in platinum-resistant ovarian cancer, per final ROSELLA trial data.

The FDA issued a CRL to RP1 plus nivolumab in advanced melanoma, accepted a NDA for the TLX101-Px imaging agent in glioma, and more.