Society for Immunotherapy of Cancer | Strategic Alliance Partners
Latest from Society for Immunotherapy of Cancer
November 14, 2020 - A novel combination comprised of the TKI cabozantinib plus the PD-1 inhibitor nivolumab and the CTLA-4 inhibitor ipilimumab is under exploration in treatment-naïve patients with intermediate or poor risk advanced or metastatic renal cell carcinoma in the phase 3 COSMIC-313 trial.

Ira Winer, MD, PhD, discusses the tolerability of ALKS 4230 plus pembrolizumab in ovarian cancer.
November 14, 2020 – Treatment with the novel intratumoral immuotherapy agent PVSRIPO resulted in an overall response rate of 33% among all patients with anti–PD-1 refractory melanoma.
November 13, 2020 - Treatment with the combination of eganelisib, a selective PI3K-γ inhibitor, and nivolumab demonstrated encouraging clinical activity and a favorable safety profile in patients with head and neck squamous cell carcinoma.

Francesca Aroldi, discusses the early efficacy data with RP2 in advanced solid tumors.
Daniel J. George, MD, discusses the expanding role of cabozantinib in renal cell carcinoma.
November 13, 2020 – The addition of urelumab, an anti-CD137 agonist to the GVAX vaccine and nivolumab led to a 30% pathologic response rate and was associated with mild toxicity as neoadjuvant or adjuvant therapy in patients with resectable pancreatic ductal adenocarcinoma.

November 13, 2020 - ALX148 showcased favorable safety and elicited objective responses when combined with trastuzumab, pembrolizumab, and multiagent chemotherapy regimens in patients with gastric/gastroesophageal junction cancer and head and neck squamous cell carcinoma.
November 13, 2020 - The combination of bempegaldesleukin and the PD-1 inhibitor nivolumab elicited a confirmed best overall response rate of 53% in efficacy-evaluable patients with metastatic melanoma.
November 13, 2020 - The genetically modified herpes simplex-1 oncolytic RP2 demonstrated encouraging clinical activity and an acceptable safety profile in heavily pretreated patients with advanced solid tumors

November 12, 2020 - The first-in-class, next-generation, DuoBody-PD-L1×4-1BB bispecific antibody GEN1046 demonstrated promising early activity and an acceptable safety profile in in patients with advanced solid tumors.
November 12, 2020 - AMG 757, a DLL3-directed, half-life extended bispecific T-cell engager, was shown to have a feasible adverse effect profile in patients with small cell lung cancer.
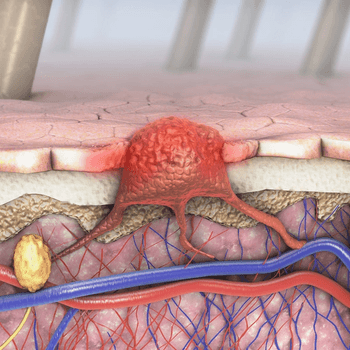
November 12, 2020 - In an effort to further improve outcomes without sacrificing tolerability, the combination of cabozantinib with pembrolizumab is being evaluated in patients with advanced melanoma in a phase 1b/2 study.

Michael B. Atkins, MD, deputy director, Georgetown-Lombardi Comprehensive Cancer Center, professor of oncology and medicine, Georgetown University School of Medicine, discusses sequencing of immunotherapy combination regimens for patients with renal cell carcinoma (RCC).
Lisa H. Butterfield, PhD, professor of medicine, surgery, and immunology, director, University of Pittsburgh Immunologic Monitoring and Cellular Products Laboratory, University of Pittsburgh, president of the Society for Immunotherapy of Cancer, discusses the potential role for immunotherapy in the treatment of patients with sarcoma.

Treatment with pembrolizumab could elicit long-term survival rates of 21% to 25% for previously-treated patients with PD-L1–positive non–small cell lung cancer compared with 3% to 4% for docetaxel.

New guidelines involving the immunotherapies nivolumab (Opdivo) and interleukin-2 inhibitors have been issued by The Society for Immunotherapy of Cancer to help practicing clinicians manage patients with renal cell carcinoma.
Lung cancer alone accounts for 26.5% of all cancer-related mortalities in the United States, which is more than any other malignancy. Despite increased awareness that smoking is an important risk factor for lung cancer, which has reduced the number of smokers in the U.S., nearly a quarter of a million new diagnoses are expected in 2016.
Howard L. Kaufman, MD, chief surgical officer, associate director for Clinical Science, surgical oncologist, Rutgers Cancer Institute of New Jersey, discusses oncolytic immunotherapy talimogene laherparepvec (T-VEC; Imlygic) for treatment of patients with melanoma, as well as the agent's side effect profile.

OncLive is pleased to announce that the Society for Immunotherapy of Cancer (SITC) has joined the OncLive Strategic Alliance Partnership program, a collaborative effort to bring up-to-date clinical information to practicing oncology specialists.

Although checkpoint blockade inhibitors targeting the PD-1/PD-L1 pathway are currently dominating the oncology community's attention, there are many other exciting approaches to anticancer immunotherapy being explored in a range of solid tumors
Yvonne Saenger, MD, director of Melanoma Immunotherapy at Columbia University Medical Center, discusses ongoing research related to immunotherapy in the field of melanoma. Sanger shared this insight during an interview with OncLive during the recent Society for Immunotherapy of Cancer Cancer Immunotherapy 101 meeting in New York City.

Jedd D. Wolchok, MD, PhD, discusses two pivotal studies, their impacts, and how far the field of immunotherapy has come in melanoma.

Antoni Ribas, MD, PhD, shares exciting clinical trial results of immunotherapy agents in patients with melanoma, as well as what the future holds for the field.

























