
During the past several decades, cancer research has produced revolutionary discoveries leading to dramatic results for many patients.

Your AI-Trained Oncology Knowledge Connection!


During the past several decades, cancer research has produced revolutionary discoveries leading to dramatic results for many patients.

The LAG-3 pathway has emerged as the next target for the use of immune checkpoint inhibitors in oncology.
Maintenance therapy with niraparib significantly improved progression-free survival compared with placebo in Chinese patients with newly diagnosed advanced ovarian cancer who achieved a complete response or partial response to first-line chemotherapy.

The addition of zilovertamab to ibrutinib will be compared with ibrutinib plus placebo in patients with relapsed/refractory mantle cell lymphoma as part of the phase 3 ZILO-301 trial.

The addition of pembrolizumab to belantamab mafodotin resulted in a higher overall response rate in patients with relapsed/refractory multiple myeloma than what has been observed with the antibody-drug conjugate alone, according to results from the phase 1/2 DREAMM-4 trial.
Enfortumab vedotin-ejfv plus pembrolizumab induced encouraging responses when used in the frontline treatment of patients with unresectable locally advanced or metastatic urothelial cancer who are not eligible to receive cisplatin-based chemotherapy.

Press Release
Casey Weaver, MD, the Wyatt and Susan Haskell Endowed Chair for Medical Excellence in the Department of Pathology, was elected to the prestigious and esteemed National Academy of Sciences.

Mario Sznol, MD, explains the evaluation of efficacy and safety when examining the benefits of new immunotherapy combinations in cancer.

A panel of hematology experts discuss use of steroids in the first-line setting as well as several subsequent-line treatments that have recently received FDA approval for patients with steroid-refractory chronic GVHD, which affects up to 50% of patients.

Single-agent bomedemstat was found to improve symptom scores, bone marrow fibrosis, spleen volumes, and anemia in patients with advanced myelofibrosis, according to findings from the phase 1/2 IMG-7289-CTP-102 trial (NCT03136185) presented during the 2022 EHA Congress.

A combination comprised of selinexor (Xpovio) and ruxolitinib (Jakafi) induced rapid spleen responses at week 12 and showcased a manageable toxicity profile in patients with treatment-naïve myelofibrosis.

The American Society of Clinical Oncology and the Association of Community Cancer Centers today jointly released two resources to help research sites increase the racial and ethnic equity, diversity, and inclusion in cancer clinical trials.

Although systemic treatments are utilized for angiosarcoma, an aggressive and rare subset of a rare cancer, the efficacy achieved with these options is not as good as it needs to be. Immunotherapy may be able to address the unmet need for a systemic treatment in this disease.

The European Medicines Agency Committee for Medicinal Products for Human Use has recommended that rucaparib no longer be used as monotherapy for the third-line treatment of patients with platinum-sensitive, relapsed or progressive BRCA-mutated high-grade epithelial ovarian, fallopian tube, or primary peritoneal cancer.

Toni K. Choueiri, MD, explains significant findings from the long-term efficacy analysis of the KEYNOTE-564 study and highlights the significance of these data for the field of RCC.
The European Medicines Agency’s Committee for Medicinal Products for Human Use has adopted a positive opinion recommending the conditional marketing authorization of teclistamab for the treatment of adult patients with relapsed/refractory multiple myeloma who have received at least 3 prior therapies, including an immunomodulatory agent, a proteasome inhibitor and an anti-CD38 antibody, and whose cancer has worsened since receiving the last treatment.

The FDA has accepted and granted priority review to a supplemental biologics license application for the use of trastuzumab deruxtecan in the treatment of adult patients with unresectable or metastatic HER2-low breast cancer who have received 1 prior therapy in the metastatic setting.

Press Release
UC Davis cancer researchers hope new technology will help diagnose and treat melanoma more successfully.

Twelve years after key research into immune checkpoint inhibitor therapy first made a splash at the American Society of Clinical Oncology Annual Meeting, evidence continues to mount that supports the durability of these agents in a range of cancers.
The American Society of Clinical Oncology and the Association of Community Cancer Centers have issued a joint statement outlining 6 recommendations for boosting participation among underserved communities.
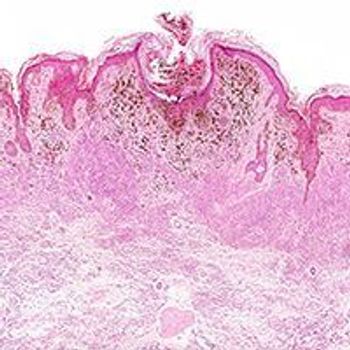
The European Medicines Agency’s Committee for Medicinal Products for Human Use has recommended the approval of the fixed-dose combination of nivolumab and relatlimab for the frontline treatment of adult and adolescents who are 12 years of age or older with advanced melanoma and a tumor cell PD-L1 expression of less than 1%.

Mutations in the p53 gene are associated with poor prognosis for patients with mantle cell lymphoma. Although modern regimens have improved outcomes, those treatments are associated with significant toxicity and, so far, have produced limited efficacy.

An influx of bispecific T-cell engagers, CAR T-cell therapies, and antibody-drug conjugates have revolutionized the treatment of hematologic malignancies; however, with several options in the sandbox, accessibility and unexplored clinical questions present challenges for optimal integration of these options into treatment.

Most patients with diffuse large B-cell lymphoma do not derive significant benefit from treatment with autologous stem cell transplantation and better therapeutic options are currently available for this population.
Zanubrutinib demonstrated a significant improvement in progression-free survival compared with orelabrutinib in patients with relapsed/refractory chronic lymphocytic leukemia and small lymphocytic lymphoma or mantle cell lymphoma.

Bradley J. Monk, MD, FACS, FACOG, discusses the importance of helping patients make informed decisions about their care options and the future role of antibody-drug conjugate targets in the biomarker testing arena.

Julie R. Brahmer, MD, MSc, discusses how data from the CheckMate 227 trial may affect the treatment landscape for NSCLC and notes the quality-of-life outcomes reported with nivolumab and ipilimumab.

The FDA has granted an orphan drug designation to DSP-0390 for the treatment of brain cancer.

Joyce O'Shaughnessy, MD, outlines the current role and ongoing research of CDK4/6 inhibitors in the treatment of patients with hormone receptor–positive, HER2-negative early stage breast cancer.

CAR T-cell therapy, autologous stem cell transplant, and novel agents each have a role to play in the second-line management of patients with primary refractory diffuse large B-cell lymphoma, according to Jason Westin, MD, MS, FACP, and Laurie H. Sehn, MD, MPH.