First-Line Options in mCRC
Axel Grothey, MD, and John Marshall, MD, discuss first-line treatment options in patients with metastatic colorectal cancer.
Episodes in this series

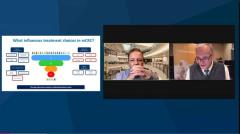
Axel Grothey, MD: There’s a breakdown that I like to see of the different types of treatments we could use right now. Fluoropyrimidine plus bevacizumab is an active treatment, and it’s definitely 1 of the treatment options I could consider in patients with low-volume disease or smoldering disease, or in elderly patients. And then anything goes in terms of doublet chemotherapy plus bevacizumab [BEV], triplet chemotherapy plus BEV. Then you see the EGF [epidermal growth factor] receptor antibodies, which have some sidedness restriction and some molecular restriction. We really need to have the molecular profile and information about sidedness available.
There’s the old standard, chemotherapy plus biologic agent. We need to pick our poison regarding the intensity of treatment according to what we want to achieve. I, for instance, see if there is a left-sided tumor, RAS, RAF, wild type, or if the patient is threatened by a tumor load and we need to force a response. I also need to see if the patient is still in decent performance status. But we know it’s going downhill very soon. I use FOLFOXIRI [fluorouracil, leucovorin, oxaliplatin, and irinotecan]. Cetuximab has an adverse effects. I’ve used FOLFOXIRI triplet plus panitumumab, and this is based on data from the German study that show when using this approach, you have a 90% response rate. And it is remarkable that we have tools in our hands where we can really, almost reliably, predict that this patient will have a response.
And then for more recently MIS [microsatellite instable]-high tumors, we have seen randomized data from KEYNOTE177 [NCT02563002] that showed pembrolizumab, a single agent, is superior to standard doublet chemotherapy plus a biologic in MSI-high mismatch repair deficient cancer. That is definitely 1 of the treatment options we can consider in first-line therapy.
Eventually, I hope we’ll have this BEACON regimen combination with BRAF inhibitor, plus an EGF receptor antibody, also moving to frontline treatment in patients with BRAF V600E-mutant tumors. It would be potentially not in the biologic, in a pure biologic doublet, but in combination with chemotherapy. That is currently being investigated. But you see the granularity in the person with aspect of first-line treatment.
John Marshall, MD: In adjuvant therapy, we are embracing the idea that capecitabine may be a better drug in the adjuvant setting; not by a lot, but by a little. I do like to use a lot of capecitabine. It’s FOLFOX [folinic acid, fluorouracil, and oxaliplatin] this, FOLFIRI [folinic acid, fluorouracil, irinotecan] that shouldn’t we be using more CAPE [capecitabine], or is it OK to use more CAPE in the metastatic setting and not just the maintenance window? What’s your thought on that?
Axel Grothey, MD: There are data and the perception that capecitabine is not tolerated well. US patients had poorer tolerability than the European and Asian patients, in particular.
And potentially, based on the folate enrichment and fortification that we have in our food, it’s because folate makes a leucovorin activated. Folate makes capecitabine more toxic.
On the other hand, now we’ve used capecitabine, CAPOX [capecitabine, oxaliplatin] in the adjuvant setting, more because you can get away with a shorter deletion of treatment compared with FOLFOX. Patients might actually lose their fear of the adverse effects. We use it commonly with radiation therapy. I have started using more CAPOX in my clinical practice, with a biologic. And there are not that great data on CAPOX plus EGF receptor antibody; that’s the 1 caveat that I have, the skin toxicity potential. But I believe, I agree. We use capecitabine a lot in the maintenance setting. We should potentially use it in combination with oxaliplatin, at least in the metastatic setting.
John Marshall, MD: I agree with that; many of us are fixed on the pump, but with FOLFOXIRI we’re still stuck with the pump, and I think when you’re using oxaliplatin it’s a perfectly legitimate alternative.
Axel Grothey, MD: Right. And you know that’s another interesting point. People think you can’t deliver oxaliplatin through a peripheral line, which is not true. You actually can do that, you just need to educate patients about it, and you need to educate your chemotherapy nurse that this can be done.
John Marshall, MD: Everybody needs a port.
Axel Grothey, MD: Right. And the moment we walk into a cancer center, the first thing the patient should get is a port.
John Marshall, MD: I’ve been leaning lately toward the triplet because of the FOLFOXIRI studies that have been done with an overall survival or a response rate, a PFS, [progression free survival] even in places where lines of therapy are all available. And I come back to, is it depth of response? What’s the reason for that? But the more heavily I’m weighing maintenance therapy, the more I lean toward FOLFOXIRI being a standard of care for a lot of patients, not just these high-risk patients. Is that too far?
Axel Grothey, MD: No, it’s not too far. I have been using triplets more than before. I believe we lost the fear of these triplets by using them in pancreas cancer. But it means for patients more poor performance status commonly, and we use it quite commonly. In our institution we kind of merge FOLFOXIRI and FOLFIRINOX into 1 regimen.
You know the subtle differences: irinotecan and bolus here, or not. We use a nonbolus 5FU containing regimen, just pure infusional 5FU. With irinotecan 150 mg and oxaliplatin 85 mg, it’s very well tolerated.
John Marshall, MD: And then the third question, you know it’s this right-left, and it’s a bit provocative. But with BRAF suddenly, if you have a mutated V600E, right/left doesn’t matter anymore. Because you’re going to use an EGFR with a BRAF in a right-sided tumor, right? So right/left goes out the window if you have V600E.
Axel Grothey, MD: If you have a V600E, we’re not there yet that I would say it’s similar to HER2 in breast cancer, where initially when you heard HER2-positive tumor and we didn’t target HER2, it was a poor prognostic marker. And now it turns into a good prognostic marker, a patient with HER2 and amplification live longer, but BRAF is still bad.
John Marshall, MD: But we’ve been saying, “Don’t use EGFR.”
Axel Grothey, MD: No, that would be in the context of a BRAF inhibitor.
John Marshall, MD: I would say right-sided, but if it’s BRAF.
Axel Grothey, MD: Correct. It serves a different purpose. I mean, it still doesn’t work with chemotherapy, but it works to counteract the feedback when you inhibit BRAF with encorafenib for another BRAF medicine. It’s actually pretty cool translation biology that they used to figure that out.
John Marshall, MD: I know, I think so, too; it’s very clever.
Axel Grothey, MD: I try to make it clear, bevacizumab is not for everyone. We talked about single-agent capecitabine, plus or minus biologic triplets, with or without a biologic.
There’s lots that we can do and there is a lot we can personalize, even beyond going for molecular markers. And the option goal is making all drugs available to all patients, not just folks in a first-line treatment, but over time looking at this continuum of care to extend lives and maintain quality of lives as long as possible.
This is what I tell my patients. That is my catchphrase on this slide here: using the least amount of treatment necessary to control their disease. And that means sometimes you do induction maintenance therapy approaches. You give patients a break from treatment. You use all the armamentarium of treatment approaches that we have available. And that’s something that is the art of oncology, helping keep patients alive and maintain their quality of life.
Transcript Edited for Clarity