Therapy After Progression in mCRC
Experts in metastatic colorectal cancer review therapeutic options after progression for patients with mCRC.
Episodes in this series

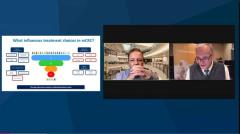
John Marshall, MD: Let’s talk about REGO [regorafenib] specifically. I’m sure everybody listeningtonight is familiar with the compound. As with all things, it works better in GI [gastrointestinal cancers] if you put a fluorine on it. It’s a multitargeted tyrosine kinase inhibitor. It hits, as I like to say, 19 different targets, sort of a dirty bomb. We’re never sure which target it’s hitting or if it’s multiple, but I also like that intellectually because I think about the targets within colorectal cancer as not being particularly strong drivers, right? Unlike say breast cancer or lung cancer, where if you find one of those targets, you totally change that patient’s outcome for the better, our drivers are OK but not that strong. Hitting more than 1 pathway at once to me, intellectually, makes sense. And this drug certainly has been proven in a variety of clinical trials. One of lead authors of the CORRECT study is on the call with me tonight, so I won’t get too gritty on that one and the CONCUR study.
You ran these studies and they were demonstrations of, to me, some very important things. One is that there were a lot of these patients around. The trials accrued quickly. The patients in this setting from around the world were willing to randomize to a placebo arm 2:1, but still a placebo arm. And it demonstrated clinical benefit and survival. The keys to the studies, when I look back, is that the patients had to be eligible, they had to be in a good performance status. They had to have good organ function. Nobody was in the hospital considering hospice or next-line therapy. They were in our clinics, driving themselves, so they were good performance patients with colorectal cancer, and we know there are plenty of them out there.
One of the places where we got lost is we then saw it as a drug that might lift somebody back from a rapidly falling performance status, and it just doesn’t do that. These drugs don’t do that. To me it’s about replicating that scenario that you led in the study about performance status, good organ function, and being able to comply. Then we’ll talk in a minute about dosing. But what’s your take on; when you were designing the studies and running them, did you think that you could pull it off?
Axel Grothey, MD: I was quite comfortable and convinced that we could pull it off. It was as you highlighted, it was one of the first, I think the first, phase 3 study where you had a placebo control in a third-line setting with a 2:1 randomization. We were wondering, and people were afraid, would the study ever accrue, because you had a placebo onboard. But they have such an unmet need. Patients wanted a new treatment because there was no other treatment.
The study accrual was projected to take 26 months. The study accrued in 10 months—16 months ahead of schedule. It shows you the unmet need that we had. And for me every patient with cancer represents an unmet need. We need to do better. We need to do warp speed for our cancer drugs for our patients.
But in the end you’re right. The drug is active. We have phase 3 data, actually 2 phase 3 studies. I also believe that less heavily pretreated patients really are the ones who should be targeted. I strongly believe not every patient with colorectal cancer needs regorafenib. Because if you have a PS2 [performance status of 2], let’s say rapidly progressing disease, deteriorating performance status, this is not the drug that can turn things around. There’s no Lazarus effect with regorafenib.
We’ve learned this over time, sometimes in a painful way, that finding the right patient is important. That’s exactly the patients that you mentioned. And fortunately, I believe, in a third-line setting, there are still a lot of patients who have good performance status, who are able to tolerate, and have adequate liver function, etc. This is a benefit of colorectal cancer, not pancreas cancer, gastric cancer. Patients in a third-line setting with gastric cancer, pancreas cancer are much sicker than in colorectal cancer on average.
John Marshall, MD: Yes, there are plenty of windows for this and TAS-102 [trifluridine, tipiracil]. We don’t think about them. If you’re considering an early phase clinical trial and you need some washout, or a rebiopsy, or a break, or you’re waiting on a slot, this is a perfect window to try some of these. I’ve been sandwiching this around liver-directed therapy on occasion when they’ve already had a lot of fluoropyrimidine.
It takes work. This is not an easy drug to administer or monitor. Don’t think, “Oh, I’m sending you away, and I’ll see you back in a month.” But if you do it and you use it correctly, you can see more clinical benefit for our patients.
What really burned this medicine early is that we used it in the wrong patients as you describe.
Transcript Edited for Clarity