Global Oncology
Latest News
EU CHMP Issues Positive Opinion for Perioperative Pembrolizumab/Enfortumab Vedotin in Cisplatin-Ineligible MIBC
Latest Videos

Shorts
Podcasts
More News
Experts in a range of breast cancer subtypes highlight research data being presented at the 2026 ESMO Breast Cancer Congress.

Data from a real-world analysis demonstrated the efficacy and safety of CAR T-cell therapy in primary mediastinal B-cell lymphoma.
Did you catch all of this week's top oncology news? Test your knowledge with OncLive's Weekly News Quiz.

Tafasitamab in combination with rituximab and lenalidomide has been approved for relapsed or refractory follicular lymphoma in Australia.
Tovorafenib has received conditional marketing authorization in Europe for patients at least 6 months of age with BRAF-positive relapsed/refractory pLGG.

China’s NMPA approved belantamab mafodotin plus Vd for the treatment of relapsed/refractory multiple myeloma after at least 1 prior line of therapy.
Did you catch all of this week's top oncology news? Test your knowledge with OncLive's Weekly News Quiz.

Zevor-cel produced durable responses in patients with relapsed/refractory multiple myeloma following at least 3 prior lines of therapy.

A BLA has been accepted in China seeking the approval of second-line trastuzumab pamirtecan for unresectable or metastatic HER2-positive breast cancer.

Catch up on insights into the global development of breast cancer therapies that are making waves around the world.
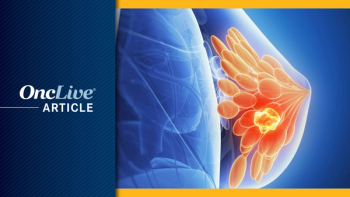
Read our recap of top phase 1 and 2 breast cancer data announcements and highlights that may influence further clinical research and developments.
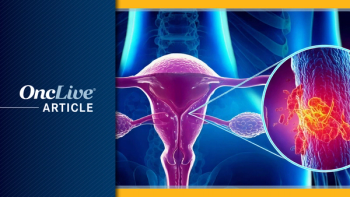
Pembrolizumab plus paclitaxel, with or without bevacizumab, is approved in the EU for pretreated platinum-resistant ovarian cancer with PD-L1 expression.

Neoadjuvant anbenitamab plus albumin-bound docetaxel yielded a significant tpCR benefit vs standard therapy in HER2-positive early breast cancer.

Lurbinectedin plus atezolizumab received CHMP recommendation as first-line maintenance for ES-SCLC that has not progressed following induction therapy.
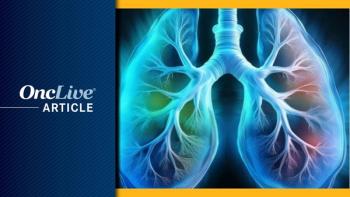
The EMA's CHMP recommends marketing authorization for tarlatamab monotherapy for ES-SCLC following relapse during or after platinum-based chemotherapy.

Neoadjuvant T-DXd followed by THP has received conditional approval in China for the treatment of patients with HER2-positive stage II/III breast cancer.
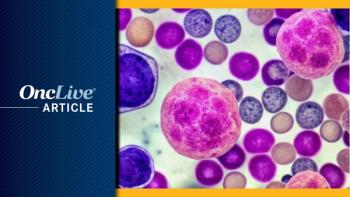
The EMA’s CHMP has recommended the approval of subcutaneous isatuximab via on-body injector for the treatment of multiple myeloma.
Subcutaneous daratumumab was approved in Europe for patient or caregiver administration in multiple myeloma.

The PI3Kα inhibitor risovalisib has been approved in Japan for PIK3CA-mutated ovarian clear cell carcinoma after progression on chemotherapy.

Read on to see what abstracts experts in the field of lung cancer are interested in learning more about at ELCC 2026 in Copenhagen, Denmark.

The EC approved an indication extension for niraparib and abiraterone acetate dual-action tablets plus prednisone/prednisolone with ADT in BRCA1/2-mutated mHSPC.

The EMA has received a Type II variation application seeking the approval of teclistamab for relapsed/refractory multiple myeloma after 1 line of therapy.

Pirtobrutinib has been approved for the treatment of chronic lymphocytic leukemia and small lymphocytic lymphoma in China.

The bispecific ADC iza-bren elicited PFS and OS benefits compared with chemotherapy in pretreated, unresectable, locally advanced or metastatic TNBC.
The European Commission has approved nogapendekin alfa inbakicept plus BCG in BCG-unresponsive NMIBC with CIS, with or without papillary tumors.