
The FDA has granted a fast track designation to the multigenic autologous CAR T-cell therapy PRGN-3006 for use as a potential therapeutic option in patients with relapsed or refractory acute myeloid leukemia.

Your AI-Trained Oncology Knowledge Connection!


The FDA has granted a fast track designation to the multigenic autologous CAR T-cell therapy PRGN-3006 for use as a potential therapeutic option in patients with relapsed or refractory acute myeloid leukemia.

Press Release
Florida Cancer Specialists & Research Institute launched a new website to host a variety of top articles ranging from recent oncology and hematology studies to new therapies, even topics on value-based care.

“I can’t conceive of anything worse than being in a situation where you can’t ask, ‘How do we get better?’ ”

Maurie Markman, MD, discusses the obstacles between implementing clinical trial data into practice.

Press Release
Results from first-of-its-kind Mount Sinai study show that immunotherapy before liver cancer surgery can kill tumor, and likely residual cancer cells.
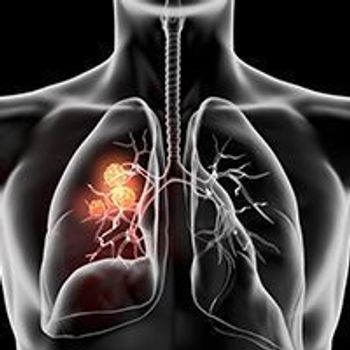
Selpercatinib continued to provide clinically meaningful benefit to patients with metastatic RET fusion–positive non–small cell lung cancer.

The FDA has approved axicabtagene ciloleucel for the treatment of adult patients with large B-cell lymphoma that is refractory to first-line chemoimmunotherapy or relapses within 12 months of first-line chemoimmunotherapy.

Evan B. Shereck, MD, MEd, talks about working with families experiencing incredible stress, the importance of wellness for both physicians and their partners, and making medical learning fun.

The expansion of the treatment armamentarium has emphasized the importance of genetic testing in patients with metastatic colorectal cancer, according to Christopher Lieu, MD, who added continued developments in the field have produced additional treatment regimens across patient subsets.

Distinctions in histology, molecular profiles, and tumor location have set diverging course of care for the treatment of patients with gastrointestinal cancers.

A biologics license application seeking the approval of 131I-omburtamab in the treatment of pediatric patients with central nervous system/leptomeningeal metastases from neuroblastoma has been resubmitted to the FDA.

Press Release
Medical oncologists Dr. Ajaz Khan, Dr. Ryan Engel, and Dr. Nitika Sharma are the latest board-certified, fellowship-trained physicians who have chosen to practice medicine at Cancer Treatment Centers of America®.

For Dawn Hershman, MD, MS, research and medicine are art forms that she uses to uplift and inspire every day.

Investigators at the Perelman School of Medicine at the University of Pennsylvania have recently initiated a clinical trial evaluating a novel CAR T-cell therapy in an effort to fill an unmet need for systemic treatment for patients with advanced incurable medullary thyroid cancer.

The addition of relacorilant to nab-paclitaxel improved overall survival compared with nab-paclitaxel alone in patients with recurrent, platinum-resistant ovarian cancer.

The newer generation antibody-drug conjugate patritumab deruxtecan has elicited encouraging responses with acceptable safety in patients with EGFR TKI–resistant, EGFR-mutated non–small cell lung cancer—a difficult-to-treat population with an unmet medical need.

The combination of eftilagimod alpha and pembrolizumab was found to produce an encouraging early overall survival rate of 73% at the 6-month landmark when used as second-line treatment in patients with PD-1/PD-L1–refractory, metastatic non–small cell lung cancer, according to data from part B of the phase 2 TACTI-002 trial.
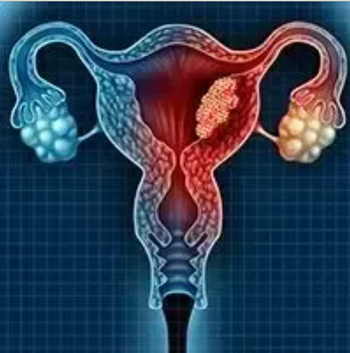
Investigators are hoping to dramatically improve the platinum-resistant ovarian cancer treatment paradigm with the development of the first-in-class antibody-drug conjugate upifitamab rilsodotin.

Rucaparib significantly improved investigator-assessed progression-free survival over placebo when used as maintenance treatment in newly diagnosed patients with advanced ovarian cancer following successful first-line treatment with platinum-based chemotherapy, according to top-line findings from the monotherapy arm of the phase 3 ATHENA trial.

Margarett Ellison, MD, gynecologic oncologist with Gynecologic Oncology of Tallahassee, A Division of Florida Cancer Specialists & Research Institute, has been designated as a National GYN Oncology Observation Site for Intuitive Surgical.

Sara A. Hurvitz, MD, FACP, led a panel of breast cancer experts—Aditya Bardia, MD, MPH; Patrick Neven, MD, PhD; and Kevin Kalinsky, MD, MS—in a review of key data from the 2021 San Antonio Breast Cancer Symposium in ER-positive breast cancer.

Shaji K. Kumar, MD, discusses key takeaways from each presentation on chronic lymphocytic leukemia, myelofibrosis, relapsed/refractory diffuse large B-cell lymphoma, CAR T-cell therapy, and multiple myeloma.

The addition of the anti-TIGIT immunotherapy tiragolumab to atezolizumab plus carboplatin/etoposide failed to significantly improve progression-free survival over atezolizumab/chemotherapy alone when used in the frontline treatment of patients with extensive-stage small cell lung cancer, missing the co-primary end point of the phase 3 SKYSCRAPER-02 trial.

Daniel J. DeAngelo, MD, PhD, discusses how the addition of uproleselan to various chemotherapy backbones is being investigated in ongoing clinical trials, plus the encouraging safety data tied to the lowered mucositis levels.

The FDA has granted priority review to a new drug application seeking the approval of futibatinib in the treatment of patients with previously treated locally advanced or metastatic cholangiocarcinoma harboring FGFR2 gene rearrangements, including gene fusions.

Next-generation spatial genomics technology paves the way for accelerating the discovery of new cancer drug targets
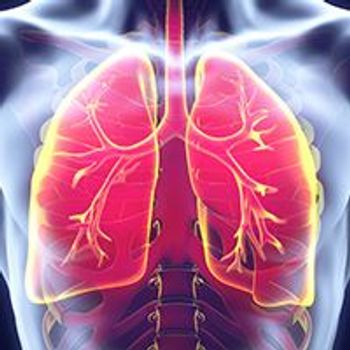
Disease progression and acquired resistance to approved EGFR tyrosine kinase inhibitors have resulted in a treatment void in the third- and later-line settings for patients with non–small cell lung cancer.

Encouraging response rates to a dual-targeted regimen in patients with recurrent or refractory malignant brain tumors with BRAF V600E mutations were reported in 2021, representing a new potential targeted therapy strategy against glioblastoma.

Sara M. Tolaney, MD, MPH, reviews practice-changing data sets throughout breast cancer and provides perspective on potential treatment approaches in the absence of definitive results.

Corey S. Cutler, MD, MPH, FRCPC, notes the clinical benefit observed with itolizumab in EQUATE and the observations that informed the design of the phase 3 EQUATOR trial.