Head & Neck Cancers
Latest News

Latest Videos

CME Content
More News
Neil D. Gross, MD, FACS, discusses the prognosis of patients who are diagnosed with cutaneous squamous cell carcinoma of the head and neck.

The European Commission has approved pembrolizumab as a monotherapy or in combination with platinum and 5-FU chemotherapy for the frontline treatment of patients with metastatic or unresectable recurrent head and neck squamous cell carcinoma and PD-L1 expression (composite positive score ≥1) on their tumors.

Nabil F. Saba, MD, FACP, discusses the potential benefit of immunotherapy in combination with radiation in head and neck cancer.
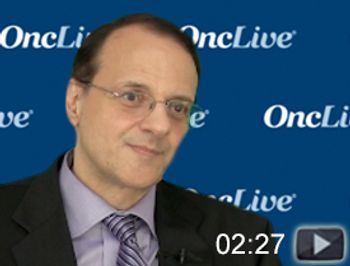
Nabil F. Saba, MD, FACP, director, Head and Neck Medical Oncology Program, Winship Cancer Institute of Emory University, and professor, Department of Hematology and Medical Oncology, Department of Otolaryngology, Emory University School of Medicine, discusses anticipated research in head and neck cancer.

Based on strong findings for efficacy and safety, as well as FDA approvals, the Society for Immunotherapy of Cancer has issued the first guidelines for the use of immunotherapy agents in patients with recurrent or metastatic head and neck squamous cell carcinoma.

The European Medicines Agency's Committee for Medicinal Products for Human Use has recommended approval of pembrolizumab as a monotherapy or in combination with platinum and 5-FU as frontline treatment for patients with metastatic or unresectable recurrent head and neck squamous cell carcinoma and PD-L1 expression on their tumors.




Jean Bourhis, MD, PhD, Radiation Oncology, Centre Hospitalier Universitaire Vaudois, discusses the rationale to explore Debio 1143 in patients with head and neck cancer.
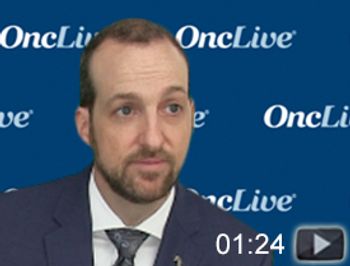
Neil D. Gross, MD, FACS, discusses a phase II study of neoadjuvant cemiplimab in patients with stage III/IV cutaneous squamous cell carcinoma of the head and neck.
Nabil F. Saba, MD, FACP, director, Head and Neck Medical Oncology Program, Winship Cancer Institute of Emory University, professor, Department of Hematology and Medical Oncology, Department of Otolaryngology, Emory University School of Medicine, discusses ongoing research in head and neck cancer.

Jean Bourhis, MD, PhD, Radiation Oncology, Centre Hospitalier Universitaire Vaudois, discusses the results of a phase II trial evaluating the addition of Debio 1143 to concurrent high-dose cisplatin and chemoradiation in high-risk patients with locally advanced squamous cell carcinoma of the head and neck.

The highly selective RET inhibitor selpercatinib (formally LOXO-292) demonstrated robust objective response rates for patients with RET-mutant medullary thyroid cancer and in those with other RET fusion-positive thyroid cancer.

The question of what the accepted therapeutic strategies for de-intensification in low-risk, HPV-related oropharyngeal squamous cell carcinoma are will be debated in an upcoming discussion.

In thyroid cancer, molecular diagnostic tests enable oncologists to evaluate thyroid nodules with atypical, suspicious, or indeterminate fine-needle aspiration cytology to determine the best course of treatment based on cytopathological result.

Vidhya Karivedu, MD, fellow/resident at the University of Cincinnati College of Medicine, discusses the antitumorigenic immune response seen with the combination of metformin and chemoradiation in patients with locally advanced head and neck squamous cell carcinoma.

Patients with differentiated thyroid cancer who were felt to not have a choice in receiving radioactive iodine treatment reportedly had lower satisfaction with the treatment decision, suggesting a need for increased shared decision making between physicians and patients.
Among patients with medullary thyroid cancer, PD-L1 positive status is linked to more aggressive clinicopathological features and predictive of structural and biochemical recurrences.

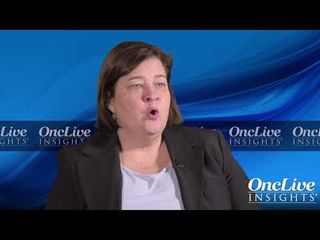
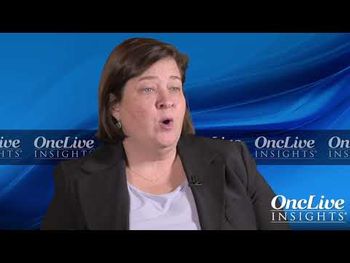
Barbara Burtness, MD, professor of Medicine (Medical Oncology); Disease Aligned Research Team Leader, Head and Neck Cancers Program; co-director, Developmental Therapeutics Research Program; of Yale Cancer Center, discusses the future of immunotherapy in head and neck cancer.
Nabil F. Saba, MD, FACP, director, Head and Neck Medical Oncology Program, Winship Cancer Institute of Emory University, professor, Department of Hematology and Medical Oncology, Department of Otolaryngology, Emory University School of Medicine, discusses investigational deintensification approaches in HPV-related oropharyngeal squamous cell carcinoma.
Nabil F. Saba, MD, FACP, director, Head and Neck Medical Oncology Program, Winship Cancer Institute of Emory University, professor, Department of Hematology and Medical Oncology, Department of Otolaryngology, Emory University School of Medicine, discusses treatment deintensification for patients with HPV-related oropharyngeal squamous cell carcinoma.
Danny Rischin, MD, director, Division of Cancer Medicine, head, Department of Medical Oncology, Peter MacCallum Cancer Centre, Melbourne, Australia, discusses the phase III KEYNOTE-048 trial, which examines pembrolizumab or pembrolizumab plus chemotherapy versus EXTREME as first-line therapy for patients with recurrent/metastatic head and neck squamous cell carcinoma.
Nabil F. Saba, MD, FACP, discussed the role of general practitioners in maintaining the health of their patients with cancer and how to better diagnosis select types of head and neck cancer.