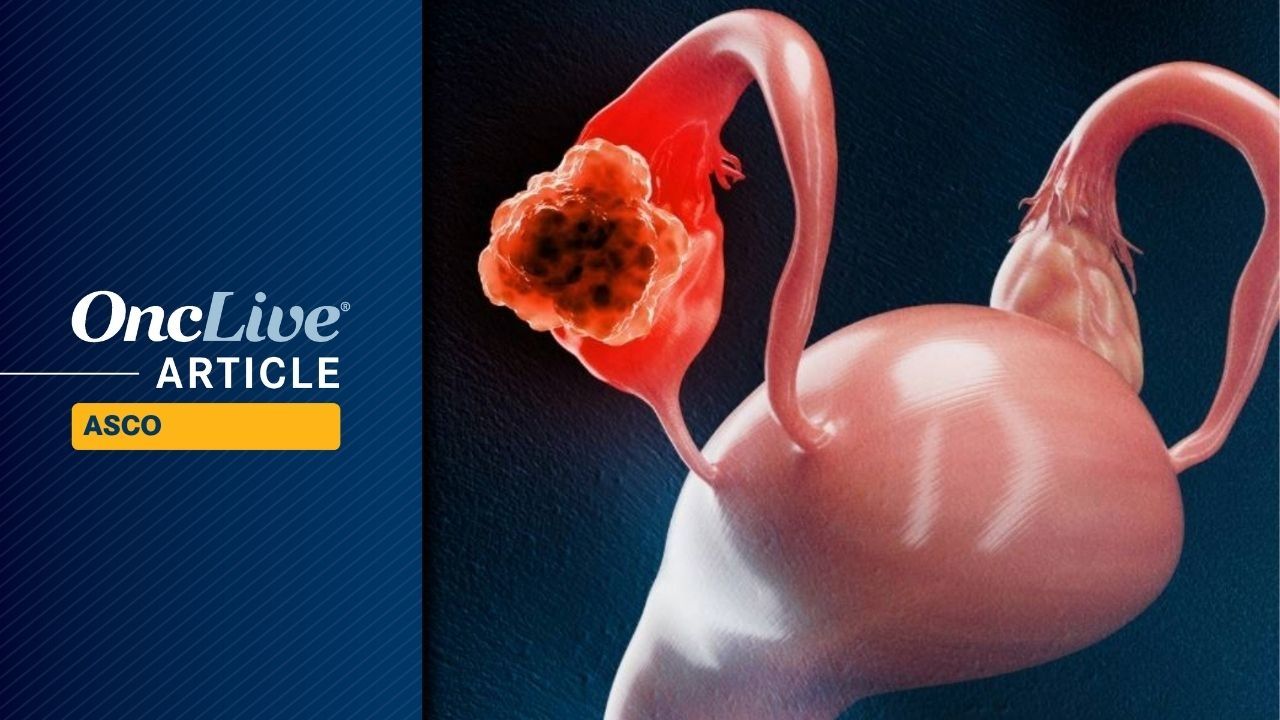
Ovarian Cancer
Latest News
Video Series

Latest Videos
Shorts








Podcasts
More News
Poll results reflect some of the most anticipated presentations and fields of interest in gynecologic oncology at the 2026 ASCO Annual Meeting.
Discover which gynecologic cancer abstracts experts believe may affect evolving treatment paradigms and patient care discussions at ASCO 2026.
AVB-001 was tolerable and activated CD8+ and CD4+ T cells without regulatory T-cell expansion in platinum-resistant high-grade serous ovarian carcinoma.

The addition of rintatolimod to pembrolizumab and cisplatin demonstrated robust albeit preliminary antitumor activity in patients with recurrent ovarian cancer.

The top 5 OncLive TV videos of the week cover insights in ovarian cancer, classical Hodgkin lymphoma, pancreatic cancer, melanoma, and myelofibrosis.

QLS5132 led to a 50% ORR and manageable safety in PROC, supporting CLDN6 as a promising ADC target in early-phase clinical development.
Early evidence suggests a dual bispecific strategy may be able to move the needle forward for patients with platinum-resistant ovarian cancer or endometrial cancer.
Experts from across oncology specialties highlight research being presented at the 2026 AACR Annual Meeting.

Mirvetuximab soravtansine plus carboplatin showed antitumor activity and a manageable safety profile in patients with recurrent platinum-sensitive ovarian cancer.
Sofetabart mipitecan elicited strong antitumor activity in recurrent platinum-resistant high-grade serous ovarian cancer.
The novel ADC DB-1311/BNT324 was effective in cervical cancer and PROC, regardless of prior treatment or histology

Single-agent rezatapopt showed efficacy and safety irrespective of patients' platinum status, prior bevacizumab or PARP inhibitor exposure, or FRα expression.
Patients who received first-line niraparib as maintenance therapy had high resolution rates and low number of discontinuations due to hematologic PSEOIs.
With 1 year of additional follow-up, avutometinib plus defactinib showed durable efficacy with no new safety concerns in LGSOC.

Results from NRG-GY019 establish chemotherapy followed by letrozole as the definitive SOC for newly diagnosed stage II-IV low-grade serous ovarian carcinoma

Relacorilant plus nab-paclitaxel cuts death risk by 35% and extends survival to 16 months in platinum-resistant ovarian cancer, per final ROSELLA trial data.

CTIM-76 received fast track designation from the FDA for pretreated platinum-resistant ovarian cancer.
Pembrolizumab plus paclitaxel, with or without bevacizumab, is approved in the EU for pretreated platinum-resistant ovarian cancer with PD-L1 expression.

Here is your cheat sheet to all therapeutic options that were cleared by the FDA in March 2026 spanning tumor types.

The FDA cleared relacorilant plus nab-paclitaxel in ovarian cancer, the primary end point of the SENTRY trial in myelofibrosis was met, and more.
Did you catch all of this week's top oncology news? Test your knowledge with OncLive's Weekly News Quiz.

The PI3Kα inhibitor risovalisib has been approved in Japan for PIK3CA-mutated ovarian clear cell carcinoma after progression on chemotherapy.

The FDA approved relacorilant plus nab-paclitaxel for previously treated platinum-resistant epithelial ovarian/fallopian tube/primary peritoneal cancer.

The top 5 OncLive TV videos of the week cover insights in chronic lymphocytic leukemia, ovarian cancer, lung cancer, breast cancer, and prostate cancer.

The FDA has cleared nivolumab/AVD for classical Hodgkin lymphoma, accepted a bezuclastinib NDA for review in nonadvanced systemic mastocytosis, and more.