
Christopher L. Moertel, MD, discusses the FDA approval of mirdametinib for patients with neurofibromatosis type 1–associated plexiform neurofibromas.

Your AI-Trained Oncology Knowledge Connection!


Christopher L. Moertel, MD, discusses the FDA approval of mirdametinib for patients with neurofibromatosis type 1–associated plexiform neurofibromas.

Neeta Somaiah, MD, highlights the evolving sarcoma treatment paradigm within the past 5 years, including the FDA approval of afamitresgene autoleucel.
Neoadjuvant chemotherapy and concurrent chemoradiation could represent a safe, effective bladder-sparing approach in MIBC.
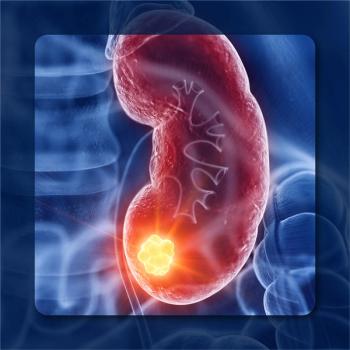
Some patients with metastatic kidney cancer achieving a good response to a combination therapy can safely stop or pause treatment after 2 years.

The FDA has accepted and granted priority review to an NDA for dordaviprone in recurrent H3K28M-mutant diffuse glioma.

Amy M. Ahnert, MD, highlights newer shared risk factors for CVD and breast cancer and questions regarding personalized screening for the diseases.
The European Commission approved belzutifan for select VHL-associated cancers and advanced clear cell renal cell carcinoma.

Tara C. Mitchell, MD, discusses considerations and lingering questions regarding pre- and post-operative management strategies in stage IIB/IIIC melanoma.

Kelly E. McCann, MD, PhD, discusses how novel ADCs and targeted therapies could improve treatment strategies in HER2-positive breast cancer.

Amy M. Ahnert, MD, details the significance of findings on the link between CVD and breast cancer as well as next steps to further define the connection.
Piflufolastat F18 imaging impacted treatment decisions and was associated with clinician confidence in prostate cancer management.

The FDA granted fast track designation to IBI363 for squamous non–small cell lung cancer.
Patients with advanced prostate cancer who initiated treatment with relugolix indicated various treatment preferences and reasons for therapy initiation.
Adherence rates improved the longer patients with metastatic and nonmetastatic prostate cancer received relugolix for a high overall adherence rate.
The FDA and European Commission have approved 2 biosimilars referencing denosumab for cancer-related bone loss and other indications.

CDK2 regulates the cell cycle and may have the potential to drive therapeutic resistance to common breast cancer drugs — including CDK4/6 inhibitors.

Christopher P. Fox, PhD, discusses the upcoming phase 3 TERZO trial of duvelisib in patients with nodal T-cell lymphoma with follicular helper T phenotype.

Ritu Salani, MD, MBA, details the significance of trials examining avutometinib plus defactinib and enthusiasm for ADCs in ovarian cancer.
Ide-cel proved feasible and effective in boosting response in patients with myeloma with suboptimal response to standard frontline therapy.
Non-myeloablative HSCT with TLI/TBI/ATG conditioning was safe and active in eliciting immune quiescence and tolerance.

CD4-, FOXP3-, and Helios-positive conventional T cells were increased in patients receiving Orca-T vs peripheral blood stem cell grafts.

The top 5 videos of the week cover insights in plexiform neurofibromas, melanoma, myeloma, leukemia, and LBCL.
No significant differences in PROs were shown between the tivozanib/nivolumab and tivozanib monotherapy arms in patients with advanced clear cell RCC.
Nivolumab in combination with cabozantinib displayed a long-term PFS benefit in patients with treatment-naive advanced RCC.

CBM588 plus cabozantinib and nivolumab shows early efficacy in metastatic renal cell carcinoma.

Efficacy results remained consistent with previous reports in the cabozantinib, nivolumab, and ipilimumab arm for patients with advanced renal cell carcinoma.

The combination of first-line avelumab and axitinib was effective and safe in a real-world analysis of patients with advanced RCC.

Updated results further supported the feasibility of VEGFR TKI interruption and immunotherapy maintenance in advanced renal cell carcinoma.
Neoadjuvant lenvatinib plus pembrolizumab followed by adjuvant pembrolizumab was safe and effective in patients with locally advanced, nonmetastatic ccRCC.
Greater responses were observed in a cohort of patients with renal cell carcinoma receiving lenvatinib plus belzutifan vs pembrolizumab plus lenvatinib.