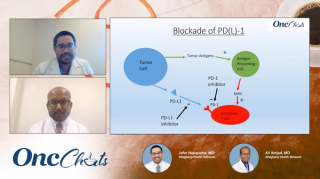
Immuno-Oncology
Latest News
Video Series

Latest Videos
More News
The efficacy of relatlimab/nivolumab was consistent across patient subgroups vs ipilimumab/nivolumab in advanced melanoma.
Sintilimab/chidamide followed by P-GemOx demonstrated preliminary efficacy in treatment-naive, early-stage extranodal natural killer/T-cell lymphoma.
Adjuvant nivolumab generated durable DFS and DMFS outcomes in resectable esophageal or GEJ cancer.

The addition of durvalumab to perioperative FLOT improved event-free survival in resectable gastric/GEJ adenocarcinoma.

Marc R. Matrana, MD, discusses the ongoing need for validated biomarkers to guide treatment selection in renal cell carcinoma.
Frontline sacituzumab govitecan plus pembrolizumab improved PFS vs chemotherapy plus pembrolizumab in PD-L1+ metastatic triple-negative breast cancer.

Here is your snapshot of all therapeutic options that were approved by the FDA in May 2025 spanning tumor types.
Ivonescimab plus chemotherapy improved PFS vs chemotherapy alone in second- and later-line EGFR-mutated non–small cell lung cancer.
Pembrolizumab/paclitaxel with or without bevacizumab improved PFS vs placebo/chemotherapy with or without bevacizumab in platinum-resistant ovarian cancer.

Aditya Shreenivas, MD, MS, discusses the FDA approval of penpulimab for recurrent/metastatic non-keratinizing NPC.
Belrestotug plus dostarlimab did not improve PFS vs dostarlimab monotherapy in PD-L1–high NSCLC, supporting the termination of belrestotug development.

The top 5 OncLive videos of the week cover insights in myelodysplastic syndrome, lung cancer, hepatocellular carcinoma, colorectal cancer, and ovarian cancer.

The FDA issues a CRL to a glioma imaging agent, an sBLA seeking approval of an epcoritamab triplet in follicular lymphoma is planned for submission, and more.

Here is your snapshot for all therapeutic options that were cleared by the FDA in April 2025 spanning tumor types.
Invikafusp alfa was active in unresectable, locally advanced or metastatic solid tumors resistant to immune checkpoint inhibitors.
Perioperative pembrolizumab with postoperative radiation with or without cisplatin showed durable EFS in resectable, locally advanced HNSCC.
Sasanlimab plus BCG significantly improved EFS vs BCG alone in high-risk non–muscle-invasive bladder cancer.

Penpulimab gets green light from FDA for nasopharyngeal carcinoma, sacituzumab govitecan combination boosts PFS in TNBC, and more.
Ivonescimab plus chemotherapy met the primary PFS end point vs tislelizumab plus chemotherapy in advanced squamous NSCLC.

Pembrolizumab/chemotherapy was approved by Health Canada for the treatment of patients with unresectable advanced or metastatic pleural mesothelioma.

The top 5 OncLive videos of the week cover insights in multiple myeloma, AL amyloidosis, myelofibrosis, breast cancer, and sarcoma.

In this episode, experts discuss the impact of neoadjuvant radiation and immunotherapy on surgical planning in patients with soft tissue sarcomas.
The PD-L1 IHC 22C3 pharmDx assay has been granted European IVDR certification for identifying pembrolizumab eligibility in gastric/GEJ adenocarcinoma.

Nerea Lopetegui-Lia, MD, discusses the prevalence of and risk factors for triple-negative breast cancer, and strategies that may improve survival outcomes.
Nivolumab-Based, Response-Adapted Approach Yields Durable Responses in Frontline Follicular Lymphoma
Response-adapted treatment with nivolumab with or without rituximab produced responses in treatment-naive follicular lymphoma.