
Results from the phase III SABRINA trial showed that subcutaneous administration of rituximab produced nearly identical response rates and toxicity as IV administration in the first-line treatment of follicular lymphoma.

Your AI-Trained Oncology Knowledge Connection!


Results from the phase III SABRINA trial showed that subcutaneous administration of rituximab produced nearly identical response rates and toxicity as IV administration in the first-line treatment of follicular lymphoma.

Shaji Kumar, MD, discusses novel therapies for patients with multiple myeloma in detail, the sequencing challenges ahead, and which current agents pose the greatest challenge.

Bradley C. Leibovich, MD, discusses the benefits of surgery and the need for more effective drug treatments for patients with metastatic renal cell carcinoma.

Tim F. Greten, MD, discussed the evolving role of immunotherapy in hepatocellular carcinoma.

Raquibul Hannan, MD, PhD, discusses the benefits of using stereotactic radiation for patients with renal cell carcinoma and highlights ongoing trials examining the modality.

Durvalumab (Imfinzi) significantly improved progression-free survival when used as a sequential treatment in patients with locally-advanced, unresectable non-small cell lung cancer who had not progressed following standard care with platinum-based chemotherapy and radiotherapy.

Fahima Dossa, MD, discussed the potential of using a watch-and-wait approach in the management of patients with rectal cancer.

The US Preventive Services Task Force announced that it recommends against screening for thyroid cancer in asymptomatic adults.

High PD-L1 expression is closely associated with local failure following radiotherapy in HPV-negative head and neck squamous cell carcinomas, according to recent study.
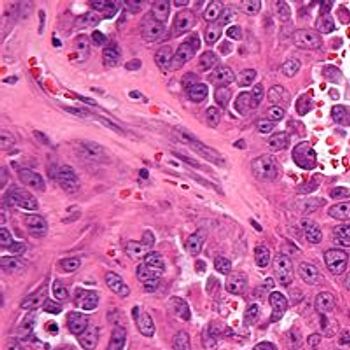
Chinese researchers have concluded that the oral VEGFR2 inhibitor apatinib demonstrated efficacy with tolerable toxicity in a small study of patients with stage IV sarcoma, according to results published in Oncotarget.

According to findings from part 2 of the phase III COLUMBUS trial, the combination of the BRAF inhibitor encorafenib and the MEK inhibitor binimetinib reduced the risk of disease progression or death by 23% compared with single-agent encorafenib for patients with BRAF-mutant melanoma.

The FDA has granted an accelerated approval to pembrolizumab for use in combination with pemetrexed plus carboplatin as a frontline treatment for patients with metastatic or advanced nonsquamous non–small cell lung cancer, regardless of PD-L1 expression.

Jonathan Strosberg, MD, discusses recent developments and emerging agents in the field of neuroendocrine tumors.

Atezolizumab missed the phase III IMvigor211 trial’s primary endpoint of improving overall survival in the second-line setting for patients with locally advanced or metastatic urothelial carcinoma.

The FDA has granted an accelerated approval to the PD-L1 inhibitor avelumab (Bavencio) for the treatment of patients with locally advanced or metastatic urothelial carcinoma with disease progression during or following platinum-containing chemotherapy, or within 12 months of neoadjuvant or adjuvant platinum-containing chemotherapy.

The FDA has approved co-packaging of the oral medications ribociclib (Kisqali) and letrozole (Femara) for the treatment of postmenopausal women with HR-positive, HER2-negative advanced breast cancer.

Wolfgang Janni, MD, PhD, discusses toxicity management with neratinib and other agents for HER2-positive breast cancer, as well as emerging treatments and impactful trials in the field.

Researchers have successfully performed autonomous retroflexion of a robotic capsule during colonoscopy in a subject animal, according to data presented during Digestive Disease Week 2017 at McCormick Place in Chicago.

The first cerebral edema death in the ZUMA-1 CAR T-cell therapy trial was disclosed today by Kite Pharma on a conference call with investors announcing the company’s first quarter financial results.

JUNO Therapeutics announced that it hopes to accelerate its process for developing CAR T cells from weeks to just 2 days.

Sattva S. Neelapu, MD, discusses the ZUMA-1 trial and the potential impact of these results on patients with non-Hodgkin lymphoma.

Christopher J. Kane, MD, discusses the challenges and impact of genetic testing on patients with prostate cancer.

Steven Coutre, MD, discusses ongoing efforts to enhance frontline outcomes for patients with CLL.

Grzegorz S. Nowakowski, MD, discusses ongoing efforts to determine prognostic factors of mantle cell lymphoma and what other challenges lie ahead.

Adding bevacizumab to standard platinum-based chemotherapy was associated with a clinically significant improvement in median overall survival in women with recurrent ovarian cancer.

Daniel Morgensztern, MD, discusses both the single-agent and combination immunotherapy data in non-small cell lung cancer and what researchers are poised to do next in the field.

Jeffrey P. Ward, MD, PhD, discusses the questions researchers still face about targeting the immune system and exactly what role T-cell receptor therapy could play in the treatment of patients with non-small cell lung cancer.

Zoptarelin doxorubicin (Zoptrex), a hybrid molecule combining an LHRH-agonist and doxorubicin, failed to improve median overall survival in a phase III advanced endometrial cancer trial, according to the manufacturer of the treatment, Aeterna Zentaris.

The European Commission has approved pembrolizumab for the treatment of adult patients with relapsed or refractory classical Hodgkin lymphoma who have progressed following autologous stem cell transplant and brentuximab vedotin, or who are transplant-ineligible and have failed brentuximab vedotin.

Researchers may be able to predict which patients with CRPC are most likely to have poorer outcomes while undergoing targeted therapy, according to the results of the PREMIERE trial.