
Himisha Beltran, MD, discusses research evaluating cell-free DNA to identify therapy resistance in prostate cancer.

Your AI-Trained Oncology Knowledge Connection!


Himisha Beltran, MD, discusses research evaluating cell-free DNA to identify therapy resistance in prostate cancer.

David P. Carbone, MD, PhD, discusses advances in immunotherapy and targeted therapy in non–small cell lung cancer and ongoing research efforts focused on acquired resistance to available therapies.

William K. Oh, MD, discusses sequencing challenges in prostate cancer, biomarker development, and the impact of the PRINT trial.

The need to establish preventive procedures that keep pace with the rapid evolution of coronavirus 2019 presents numerous challenges for the oncology field as institutions adapt standard treatment protocols to minimize the risk for virus spread among clinicians and patients alike.

Tocilizumab, a treatment used for adverse events related to cancer therapy, among other indications, is being explored in a phase III trial as a treatment for adult patients hospitalized with severe COVID-19 pneumonia, according to Genentech (Roche), the manufacturer of the interleukin-6 receptor antagonist.

Terence T. Sio, MD, MS, discusses the utility of proton therapy in lung cancer.

Olaparib has received an orphan drug designation in Japan for the maintenance treatment of germline BRCA-mutated curatively unresectable pancreatic cancer.

Topline results from the pivotal phase III FLASH trial showed that SGX301 demonstrated strong clinical activity in patients with cutaneous T-cell lymphoma.

John P. Sfakianos, MD, discusses some of the optimal and evolving management strategies in localized bladder cancer.

Eugene B. Cone, MD, discusses the advantages of the study, the need for such research, and the potential to broaden these efforts to more novel agents such as abiraterone acetate, enzalutamide, and immune checkpoint inhibitors.

The Chinese National Medical Products Administration has approved almonertinib (Ameile; HS-10296) for the treatment of patients with EGFR T790M mutation–positive non–small cell lung cancer who have progressed on or after other EGFR TKI therapy.

Physicians treating patients with cancer may want to consider postponing surgeries or altering therapy plans because of the threat that the coronavirus poses to this population.

Don S. Dizon, MD, FACP, FASCO, discusses potential challenges for patients with gynecologic cancers.

Che-Kai Tsao, MD, discusses the current unmet needs in adjuvant therapy for patients with renal cell carcinoma and how current research is addressing such challenges.

David S. Hong, MD, discusses the burgeoning role of multitargeted therapeutics in patients with hematologic malignancies.

Anastasios Dimou, MD, discusses available and emerging treatment options and approaches for patients with oncogenic-driven lung cancer.

Prithviraj Bose, MD, discusses the high unmet need in post-myeloproliferative neoplasms acute myeloid leukemia and the impact of findings from the phase I/II trial on current and future treatment strategies in the space.

The FDA has granted 2 Orphan Drug Designations to ALPN-101 for the prevention and treatment of acute graft-versus-host-disease.

Jeremy L. Warner, MD, MS, discusses the goals of the COVID-19 and Cancer Consortium Registry, what other data need to be extrapolated with COVID-19, and how the virus could impact patients with cancer.
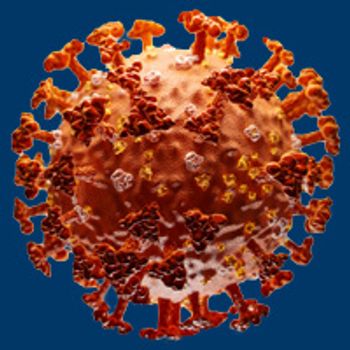
As the novel coronavirus continues its rapid global spread, many oncology-based organizations have been forced to make the difficult decision to either cancel or postpone highly anticipated medical conferences.

Chad Cherington, MD, discusses groundbreaking developments and critical next steps across the spectrum in lung cancer.

Julie N. Graff, MD, discusses the results of the KEYNOTE-199 trial, the importance of combination strategies in metastatic castration-resistant prostate cancer, and the next phase of research for the combination.

Combining durvalumab with standard frontline chemotherapies showed a sustained overall survival benefit in patients with extensive-stage small cell lung cancer, but a regimen of durvalumab, chemotherapy, and tremelimumab did not boost OS.

Balazs Halmos, MD, discusses the implications of the emergence of osimertinib in the frontline treatment of patients with EGFR-mutated non–small cell lung cancer, the efforts being made to address resistance to the third-generation EGFR TKI, and the promise of circulating tumor DNA in the space.

Neeta Somaiah, MD, discusses ongoing research in the field of sarcoma.

Jonathan C. Trent, MD, PhD, discusses recent advances and ongoing research in the field of soft tissue sarcoma.

Haploidentical allogeneic stem cell transplant is a viable treatment option for patients with myelofibrosis who have a higher risk for posttransplant complications and mortality.

Anis Hamid, MBBS, discusses the analysis of genomic profiling from the phase III CHAARTED trial, and the burgeoning potential of precision medicine in metastatic hormone sensitive prostate cancer.

The China National Medical Products Administration has accepted a supplemental New Drug Application for niraparib for use as a frontline maintenance treatment of adult patients with advanced epithelial ovarian, fallopian tube, or primary peritoneal cancer who are in a complete or partial response to first-line platinum-based chemotherapy.

Juan C. Lopez-Mattei, MD, FACC, FASE, FSCCT, FSCMR, discusses the findings of the retrospective analysis of pulmonary hypertension in patients with myelofibrosis and their impact on clinical practice.